Research Summary
Mitochondria-derived vesicles act as a first line of defence in response to mitochondrial insults, which precedes mitophagy. Our lab is interested in understanding the molecular pathways of MDV biogenesis and trafficking to other organelles. By mapping the trafficking itinerary of this class of ancient vesicles, we hope to understand how endosymbiosis forged the development of eukaryotic endomembranes. Further, increasing the mito-protective release of MDVs in cardiac diseases could stall the progress of several cardiovascular diseases characterised by mitochondrial dysfunction such as atherosclerosis, diabetic microvascular disease, ischaemia-reperfusion injury, and chronic heart failure among others.
Research interests
Mito-nuclear communication
- Mitochondrial quality control
MDV biogenesis
Regulation of intracellular trafficking
Actin regulation of MDV trafficking
Research Programs
Mitochondria, having arisen from ancient alphaproteobacteria, are suggested to have created the immense evolutionary pressure for the elaboration of an endomembrane system in the earliest eukaryotes. It is intriguing that mitochondria-derived vesicles (MDVs), discovered a decade back, might be one of the first kind of intracellular vesicles encountered in ancestral eukaryotes/archaea. Therefore, there is likely to be a great extent of unexplored crosstalk between the endomembrane system and the mitochondria that keep mitochondria healthy. We will devise novel ways to purify these ~70 nm mitochondria derived vesicles from endothelial cells and uncover their roles in blood vessel formation.Preliminary observations suggest that RhoGTPases that play a role in vesicular traffickingare nonetheless important in mitochondrial function. I would like to expand on the potential role of vesicular RhoGTPases in mitochondrial health and their relevance to endothelial metabolism and stability of the vasculature. 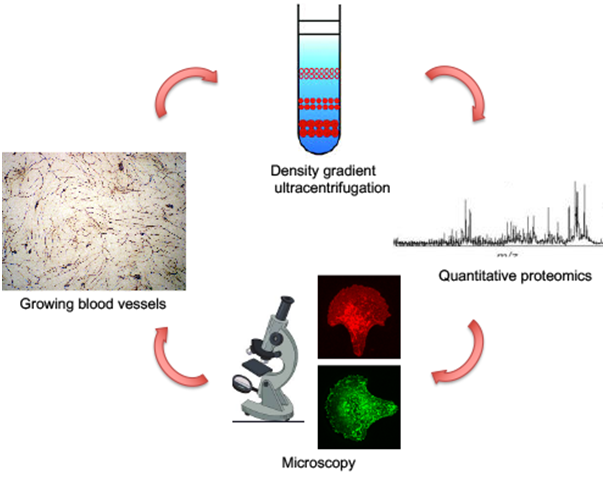
Current Research Grants
-
2025 2020
Ramalingaswami Fellowship
Department of Biotechnology [DBT] -
2025 2022
Characterising Mitochondria-derived Vesicle Trafficking through a Proximity Labelling Approach- A possible Novel Mito-nuclear Communication Pathway
SERB, Department of Science & Technology [DST]